 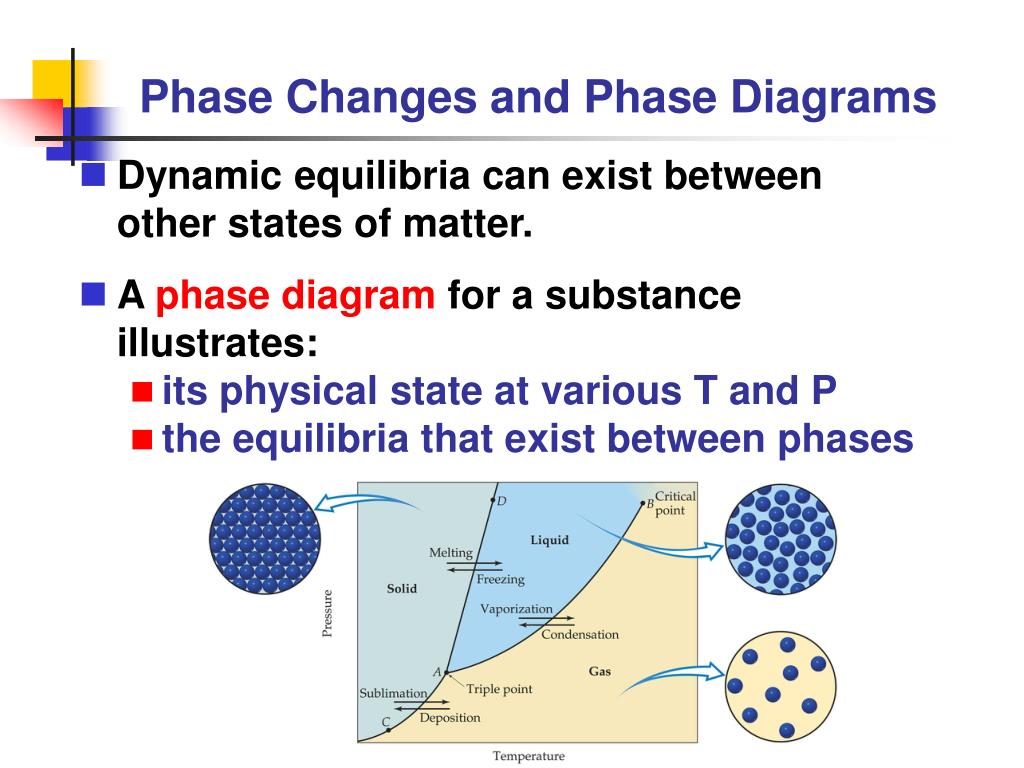
The temperature of a cup of soda and ice that is initially at 0 ☌ ☌ stays at 0 ☌ ☌ until all of the ice has melted. There is no temperature change until a phase change is completed. Similarly, energy is needed to vaporize a liquid to overcome the attractive forces between particles in the liquid. Since the energy involved in a phase changes is used to break bonds, there is no increase in the kinetic energies of the particles, and therefore no rise in temperature. Phase changes to a less energetic state are as follows:Įnergy is required to melt a solid because the bonds between the particles in the solid must be broken. Vaporization-Liquid to gas (included boiling and evaporation).Phase changes to a more energetic state include the following: (d) The atmosphere is ionized in the extreme heat of a lightning strike.ĭuring a phase change, matter changes from one phase to another, either through the addition of energy by heat and the transition to a more energetic state, or from the removal of energy by heat and the transition to a less energetic state. A gas must be held in a closed container to prevent it from moving out into its surroundings. (c) Particles in a gas are separated by distances that are considerably larger than the size of the particles themselves, and they move about freely. Water can flow, but it also remains in an open container because of the forces between its molecules. Forces between them strongly resist attempts to push them closer together and also hold them in close contact. (b) Particles in a liquid are also in close contact but can slide over one another. This rock retains its shape because of the forces holding its atoms or molecules together. These particles are essentially in contact with one another. 
This produces an ionized gas that is a combination of the negatively charged free electrons and positively charged ions, known as plasma.įigure 11.8 (a) Particles in a solid always have the same neighbors, held close by forces represented here by springs. The plasma state is reached by heating a gas to the point where particles are pulled apart, separating the electrons from the rest of the particle. Although you may not have heard much about plasma, it is actually the most common state of matter in the universe-stars are made up of plasma, as is lightning. The most energetic state of all is plasma. 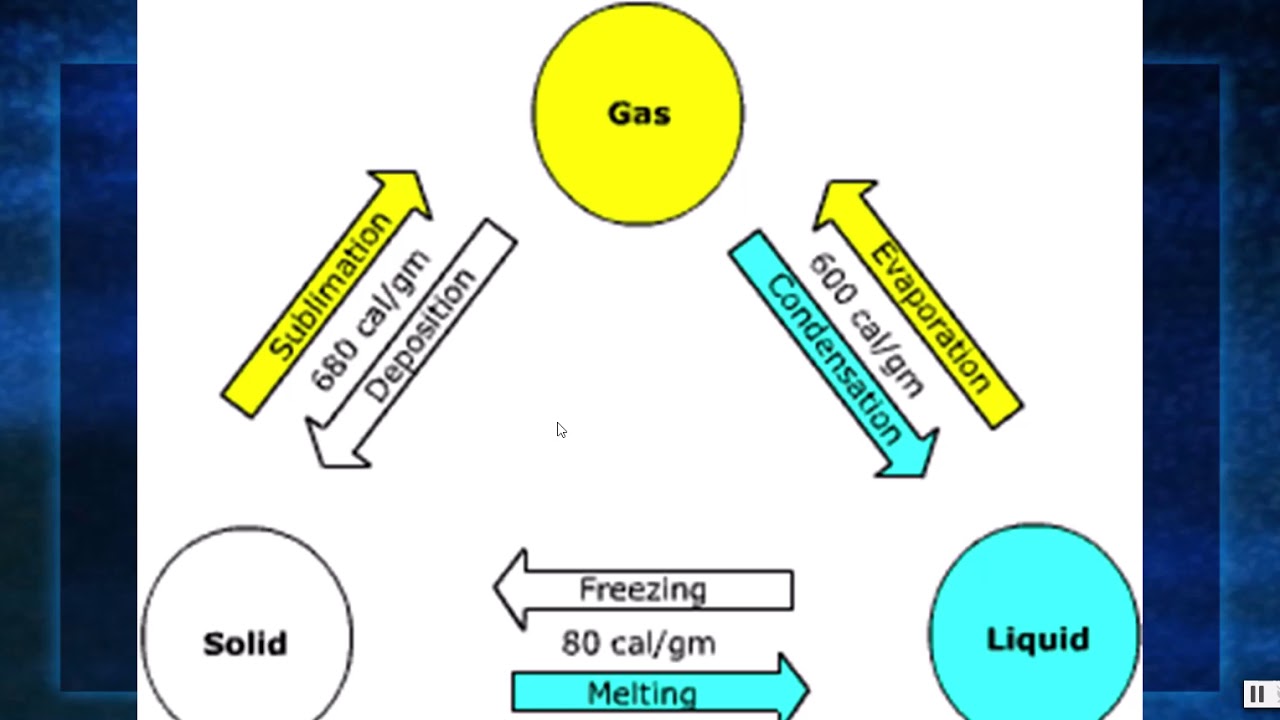
Particles in gases are separated by distances that are large compared with the size of the particles. Gas has a more energetic state than liquid, in which particles are broken free of their bonds. Liquid has a more energetic state, in which particles can slide smoothly past one another and change neighbors, although they are still held together by their mutual attraction. (These forces can be thought of as springs that can be stretched or compressed, but not easily broken.) Solid has the least energetic state atoms in solids are in close contact, with forces between them that allow the particles to vibrate but not change position with neighboring particles. 
The three phases of matter that you frequently encounter are solid, liquid and gas (see Figure 11.8). Because this energy enters or leaves a system during a phase change without causing a temperature change in the system, it is known as latent heat (latent means hidden). But surprisingly, there are situations where adding energy does not change the temperature of a substance at all! Instead, the additional thermal energy acts to loosen bonds between molecules or atoms and causes a phase change. So far, we have learned that adding thermal energy by heat increases the temperature of a substance. Introduce this section by asking students to give examples of solids, liquids, and gases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
